Answers
Answer: here u go
Explanation:
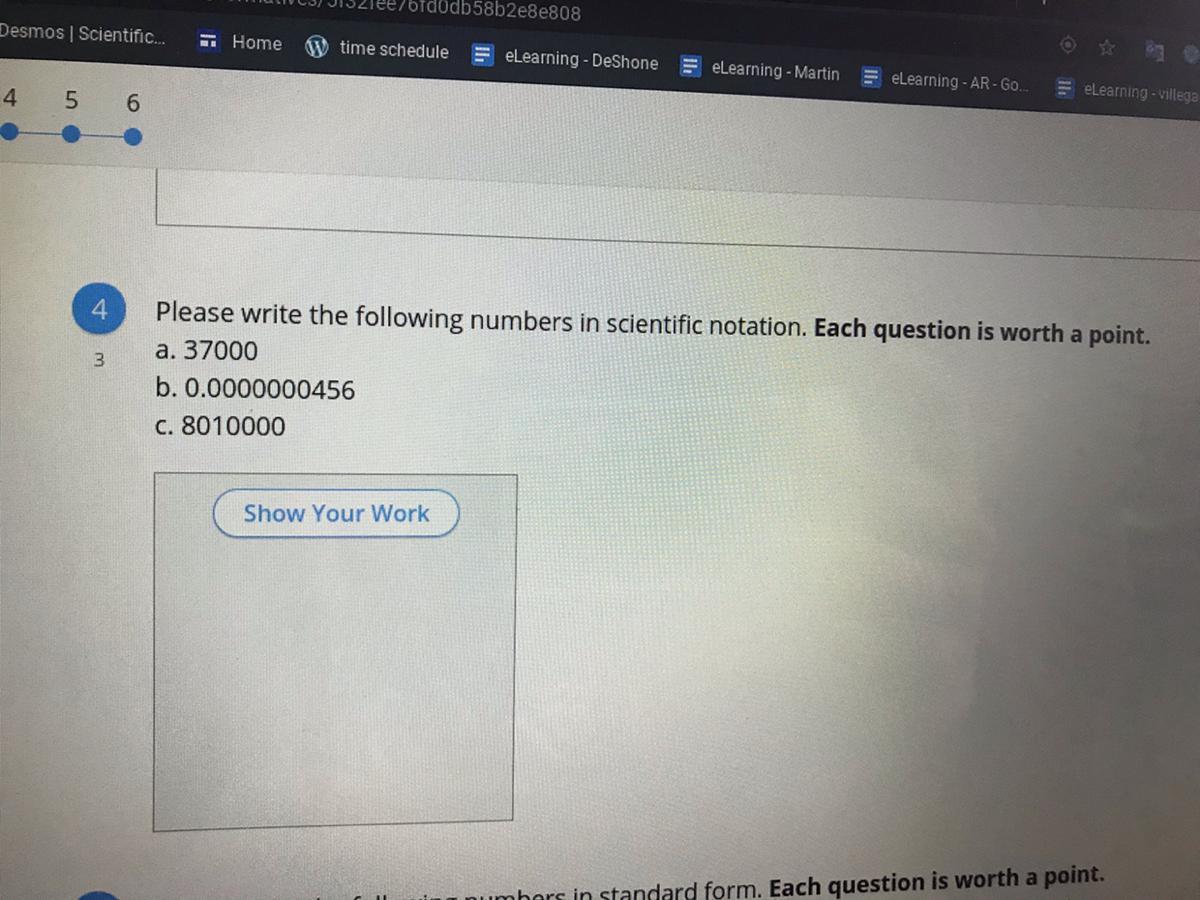
A. 3.7 x 10^4
B. 4.56 × 10^-8
C. 8.01 × 10^6
Work for A:
Step 1
To find a, take the number and move a decimal place to the right one position.
Original Number: 37,000
New Number: 3.7000
Step 2
Now, to find b, count how many places to the right of the decimal.
New Number: 3 . 7 0 0 0
Decimal Count: 1 2 3 4
There are 4 places to the right of the decimal place.
Step 3
Building upon what we know above, we can now reconstruct the number into scientific notation.
Remember, the notation is: a x 10b
a = 3.7 (Please notice any zeroes on the end have been removed)
b = 4
Now the whole thing:
3.7 x 104
Step 4
Check your work:
104 = 10,000 x 3.7 = 37,000
Work for B:
Step 1
To find a, take the number and move a decimal place to the right one position.
Original Number: 456
New Number: 0.0000000456
Step 2
Now, to find b, count how many places to the right of the decimal.
New Number: 0 . 0 0 0 0 0 0 0 4 5 6
Decimal Count: 1 2 3 4 5 6 7 8 9 10
There are 2 places to the right of the decimal place.
Step 3
Building upon what we know above, we can now reconstruct the number into scientific notation.
Remember, the notation is: a x 10b
a = 4.56
b = 2
Now the whole thing:
4.56 x 102
Step 4
Check your work:
102 = 100 x 4.56 = 456
Work for C:
Step 1
To find a, take the number and move a decimal place to the right one position.
Original Number: 8,010,000
New Number: 8.010000
Step 2
Now, to find b, count how many places to the right of the decimal.
New Number: 8 . 0 1 0 0 0 0
Decimal Count: 1 2 3 4 5 6
There are 6 places to the right of the decimal place.
Step 3
Building upon what we know above, we can now reconstruct the number into scientific notation.
Remember, the notation is: a x 10b
a = 8.01 (Please notice any zeroes on the end have been removed)
b = 6
Now the whole thing:
8.01 x 106
Step 4
Check your work:
106 = 1,000,000 x 8.01 = 8,010,000
Hope this helps!
Related Questions
What does chemical equations and chemical formulas have in common?
Answers
Answer:
Chemical symbols refer to chemical elements only. They do not necessarily refer to atoms of that element, but also to ions.
Explanation:
The term organic does not mean “natural” true or false
Answers
Organic foods are grown without artificial pesticides .
HELPP GIVING AWAY 30 POINTS!!!!!
Determine Your family wants to use
renewable energy to heat your home. Which
renewable energy resource is best suited to
your area? Explain your answer.
Answers
Answer:
wood
Explanation:
i can take the seds from the trees to row new trees and still use the wood without causing deforestation
Which of the following is NOT a strong electrolyte?
:
Answers:
Na2SO4
KI
CaCl2
LiOH
C6H1206
Answers
Answer:
C6H1206
Explanation:
C6H12O6 is a monomer of carbohydrates also known as glucose, so it is not an electrolyte at all.
In the combustion of hydrogen gas, hydrogen reacts with oxygen from the air to form water vapor. hydrogen+oxygen⟶water If you burn 58.9 g of hydrogen and produce 526 g of water, how much oxygen reacted?
Answers
Answer:
[tex]m_{O_2}=467gO_2[/tex]
Explanation:
Hello.
In this case, for the reaction:
[tex]2H_2+O_2\rightarrow 2H_2O[/tex]
The correct way to compute the oxygen that reacted is by considering the mass of hydrogen, 58.9 g, its molar mass, 2.02 g/mol, the 2:1 mole ratio between hydrogen and oxygen and the atomic mass of that gaseous oxygen 32.00 g/mol; therefore we use the following stoichiometric procedure:
[tex]m_{O_2}=58.9gH_2*\frac{1molH_2}{2.02gH_2}*\frac{1molO_2}{2molH_2} *\frac{32.00gO_2}{1molO_2} \\\\m_{O_2}=467gO_2[/tex]
The same result could have been obtained by using the mass of water since the law of conservation of mass is obeyed here.
Best regards!
A student was performing a separation of a mixture of organic compounds. The final step of the process involved a filtration of the analyte from an aqueous solution. After drying the filtered solid for a very short period time, they took the melting point of the compound. The measured melting point range of the compound was 106 – 113.8 0C, while the literature melting point of the compound was 122.3 0C. The above scenario is a very common one in organic labs.
1. Do you think their sample was pure?
2. If not, then what do you think could be the source of error.
3. How do you think this error can be minimized?
Answers
Answer:
1) No
2) The solvent contaminated the analyte
3) The solvent should be evaporated properly before washing and drying the analyte
Explanation:
During separation of organic compounds, solvents are used. These solvents are able to contaminate the analyte and lead to a large difference in melting point of solids obtained.
However, the error can be minimized by evaporating the solvent before washing, drying and melting point determination of the solid.
A sample of an unknown gas weighs 0.419 grams and produced 5.00L of gas at 1.00atm (unknown gas only) and 298.15K, what is the molar mass (g/mole) of this unknown gas
Answers
Answer:
molar mass of unknown gas = 1.987 g/mol
Explanation:
First, the number of moles of the unknown gas is found
Using the ideal gas equation: PV = nRT
P = 1.00 atm, V = 5.00 L, T = 298.15 K, R = 0.082 L.atm.mol⁻¹K⁻¹
n = PV/RT
n = (1.00 atm * 5.00 L)/(298.15 K *0.082 L.atm.mol⁻¹K⁻¹)
n = 0.2109 moles
Molar mass = mass/ number of moles
molar mass = 0.419 g/ 0.2109 mols
molar mass of unknown gas = 1.987 g/mol
The molar mass of unknown gas by using ideal gas equation = 1.987 g/mol.
Ideal gas equationThis equation gives the relation between pressure, volume, temperature as given below:
[tex]PV = nRT[/tex]
P = 1.00 atm, V = 5.00 L, T = 298.15 K, R = 0.082 L.atm.mol⁻¹K⁻¹
Substitute the above values in the above equation as follows:
n = (1.00 atm * 5.00 L)/(298.15 K *0.082 L.atm.mol⁻¹K⁻¹)
n = 0.2109 moles
Formula for molar mass[tex]Molar mass = mass/ number of moles[/tex]
Calculate molar mass by using the above equation,
molar mass = 0.419 g/ 0.2109 moles
The molar mass of unknown gas = 1.987 g/mol
Find more information about ideal gas equation here,
brainly.com/question/4147359
Please answer !! For chemistry
Answers
Answer:
a. is the correct answer
Explanation:
What is the percent error for the experiment if the actual density is
2.49g/mL but the experimental value is 1.47 g/mL?
Answers
Answer:
The answer is 40.96%Explanation:
The percentage error of a certain measurement can be found by using the formula
[tex]P(\%) = \frac{error}{actual \: \: number} \times 100\% \\ [/tex]
From the question
actual density = 2.49g/mL
error = 2.49 - 1.47 = 1.02
We have
[tex]p(\%) = \frac{1.02}{2.49} \times 100 \\ = 40.96385542...[/tex]
We have the final answer as
40.96 %Hope this helps you
A baseball strikes the roof of a car and dents it. The paint on the roof begins to crack and chip, exposing the metal. The exposed metal on the roof rusts, eventually causing a small hole in the roof. 17. Which event is a chemical change? The baseball strikes the roof The roof of the car dents The paint cracks and chips The exposed metal rusts
Answers
Answer:
The exposed metal rusts is an example of a chemical change because rust is an example of a chemical change in objects for example bicycles, scooters, etc.
Based on the visible cell structures, which of the following statements is true?
All of the cells are plant cells.
All of the cells have chloroplasts.
All of the cells are animal cells.
All of the cells have a nucleus.
Answers
What can you infer about the likely occurrence of the other isotopes in each of the
above elements? Explain your reasoning.
Answers
Answer:
Most common has the same or closest mass because it is closer to the original’s mass.
Explanation:
I got it from the answer key
Read more here:
https://brainly.com/question/1598931
Be sure to answer all parts.
Write a balanced equation for each reaction. Do not include states of matter to your equation.
H2 + O2 H20
Answers
Answer:
2H₂ + O₂ → 2H₂O
Explanation:
Chemical equation:
H₂ + O₂ → H₂O
Balance chemical equation:
2H₂ + O₂ → 2H₂O
Step 1:
H₂ + O₂ → H₂O
Left hand side Right hand side
H = 2 H = 2
O = 2 O = 1
Step 2:
H₂ + O₂ → 2H₂O
Left hand side Right hand side
H = 2 H = 4
O = 2 O = 2
Step 3:
2H₂ + O₂ → 2H₂O
Left hand side Right hand side
H = 4 H = 4
O = 2 O = 2
What is the correct 1 letter code for the amino acid sequence Glutamic Acid-Histadine-Tyrosine Select one: a. E-H-Y b. G-H-T c. Q-H-T d. E-H-T
Answers
Answer:
a. E-H-Y
Explanation:
A group of three nucleotides is called a codon that codes for a specific amino acid in the protein. There are 20 essential amino acids present in human body and are required in the diet.
Each amino acid is given a one-letter code that makes the study of amino acid sequences easy. One letter code for the given amino acid sequence Glutamic Acid-Histidine-Tyrosine is E-H-Y in which E is code for Glutamic Acid, H is a code for Histidine, and Y is a code for Tyrosine.
Hence, the correct answer is "a. E-H-Y".
CHEMISTRY!! 50 POINTS!
There are 5.5 L of a gas present at -38.0 C. What is the temperature if the volume of the gas has changed to 1.30 L?
Answers
Answer:
The answer to this question is 33.8
Which of the following evidence supports the theory of plate tectonics
Answers
Answer:
seafloor spreading
Explanation:
i took the test
If the density of aluminum is 2.7g/ml, what is the volume of 12.0g?
Answers
Answer:
The answer is 4.44 mLExplanation:
The volume of a substance when given the density and mass can be found by using the formula
[tex]volume = \frac{mass}{density} \\ [/tex]
From the question we have
[tex]volume = \frac{12}{2.7} \\ = 4.4444444....[/tex]
We have the final answer as
4.44 mLHope this helps you
20
How do you determine the number of barium atoms in the formula below?
4Ba(OH)2
F
# of Ba atoms = coefficient 4 + subscript 1 = 5
G
# of Ba atoms = coefficient 4 X subscript 1 = 4
H
# of Ba atoms = subscript 4 X coefficient 2 = 6
# of Ba atoms coefficient 1 + subscript 1 = 2
J
Answers
Answer:
G is correct option:
# of Ba atoms = coefficient 4 × subscript 1= 4
Explanation:
Formula:
4Ba(OH)₂
G is correct option:
# of Ba atoms = coefficient 4 × subscript 1= 4
Because there are only 4 atoms of Ba in given formula.
Ba(OH)₂ contain one atom of Ba while in question there are 4 moles of Ba(OH)₂ present thus total 4×1 = 4 atoms of Ba present.
Other options are incorrect. Because,
F:
# of Ba atoms = coefficient 4 + subscript 1 = 5
This shows given formula contain 5 Ba atoms. So it is incorrect.
H:
# of Ba atoms = subscript 4 × coefficient 2 = 6
This shows that subscript is 4 which is incorrect because coefficient is 4 and subscript is 1.
j:
# of Ba atoms = subscript 1 + coefficient 1 = 2
This option shows that subscript is one which is correct but coefficient is incorrect. The coefficient of Ba is 4 and coefficient is always multiply with subscript not added. So this option is also incorrect.
A species that has 13 protons and 10 electrons will be_____
Answers
Answer: Aluminum
Explanation:
Al3+ indicates an ion of aluminum having a charge of + 3. I.e., since an aluminum atom normally has 13 protons and 13 electrons, this ion has 10 electrons (-10 charge) and 13 protons (+ 13 charge) giving it a charge of + 3 (-10 + 13 = +3).
Answer:
Explanation:
Al3+ indicates an ion of aluminum having a charge of + 3. I.e., since an aluminum atom normally has 13 protons and 13 electrons, this ion has 10 electrons (-10 charge) and 13 protons (+ 13 charge) giving it a charge of + 3 (-10 + 13 = +3).
ccto.
Plz answer me will mark as brainliest
Answers
Explanation:
Valves
Blood capillaries
If 5.00g of iron metal is reacted with 0.950g of Cl2 gas, how many grams of ferric chloride (FeCl3) will form?
Answers
Answer:
1.45g of FeCl3
Explanation:
The equation of the reaction is given as;
2Fe + 3Cl2 --> 2FeCl3
2 mol of Fe reracts with 3 mol of Cl2 to form 2 mol of FeCl3
Upon converting to mass using;
Mass = Number of moles * Molar mass
( 2 * 55.85 = 111.7g ) of Fe reacts with ( 3 * 71 = 213g ) of Cl2 to form ( 2 * 162.2 = 324.4g) of FeCl3
Cl2 is the limiting reactant as it determines how much of FeCl3 is formed
213g of Cl2 = 324.4g of FeCl3
0.950g of Cl2 = x
x = (0.950 * 324.4 ) / 213
x = 1.45g of FeCl3
If 200.00 mL of a solution of sugar and water contains 0.0025 moles of sugar, what is the concentration of the solution in units of moles per unit volume? Round decimals to the nearest thousandth where applicable. Do not include trailing zeros in decimal answers (e.g., 0.05 instead of 0.050).
Answers
Answer:
0.013 mol/L
Explanation:
Molarity, which is molar concentration or moles per unit volume, can be calculated using the formula:
M = n/V
Where; M = Molar concentration of solution
n = number of moles of solute
V = Volume of solvent
n = 0.0025 moles of sugar
V = 200.00mL of solution
Since 0.001L = 1mL
200mL = 0.200L
Therefore, M = n/V
M = 0.0025/0.200
M = 0.0125 mol/L
Round up to the nearest thousandth ~ 0.013mol/L
What is the final concentration if water is added to 0.25 L of a 8M NaOH solution to make 4.0 L of a diluted NaOH solution?
.25 M NaOH solution
1 M NaOH solution
.5 M NaOH solution
Answers
Answer:
0.5 M NaOH solution
Explanation:
Initially, we have 0.25L or 250 mL of 8M NaOH solution
Which means that 1L of this solution would have 8 moles of NaOH
Moles present in 250 mL of solution:
Molarity * Volume( in L)
8 * 0.25 = 2 moles
There is 2 moles of NaOH in 0.25 L of the provided NaOH solution
More water is going to be added to this solution but the number of moles of NaOH will remain the same
now that the solution has a volume of 4L after adding the additional water, we have a 4L solution which contains 2 moles of NaOH
Molarity of the new solution:
Molarity = Number of moles in solution / Volume of solution
Molarity = 2 / 4
Molarity = 0.5 M
Question 11
4 pts
Using the formula 2H202 --> 2H2O + O2, if 7.30 moles of peroxide are
decomposed, how many moles of oxygen will be formed?
Answers
Answer:
3.65 mol O₂
Explanation:
Step 1: RxN
2H₂O₂ → 2H₂O + O₂
Step 2: Define
Given - 7.30 mol H₂O₂
Solve - x mol O₂
Step 3: Stoichiometry
[tex]7.30 \hspace{3} mol \hspace{3} H_2O_2(\frac{1 \hspace{3} mol \hspace{3} O_2}{2 \hspace{3} mol \hspace{3} H_2O_2} )[/tex] = 3.65 mol O₂
HELPPPPP
Identify whether each of the following changes is a physical change or a chemical change. Write “P” on the line for a physical change and a “C” for a chemical change.
22. Water boiling ______23. Iron rusting ______24. Butter melting _____25. Wood rotting ______
26.Alcohol evaporating _____27.Glass breaking ______28.Mowing the lawn ______29.Baking a cake ______
Answers
Answer:
22 is P
23 is C
24 is P
25 is C
26 is P
27 is P
28 is P
29 is C because youre using thermal heat and you cannot return to raw batter
Four grams of hydrogen react completely with 32 grams of oxygen. Based on the law of conservation of mass how many grams of water will be produced
Answers
Se producirán 36 gramos de agua
Explicación: Ya que en una reacción química no puede descubrir ningún cambio en masa total de las sustancias que en ella intervienen, pues en una reacción química ni se gana ni se pierde masa.
A scientist discovers remnants of an organism on a slide under his microscope. He can only identify a few components: a large vacuole, ridged cell wall, and a chloroplast. Was the organism prokaryotic or eukaryotic? How do you know?
Answers
Answer:
Eukaryotic (a plant cell)
Explanation:
The presence of a chloroplast indicates that the cell has membrane-bound organelles. This is not a feature of prokaryotic cells - only eukaryotic cells. Therefore, the cell is eukaryotic. The presence of a large vacuole, chloroplast, and rigid cell wall suggests its a plant cell as plant cells are the only eukaryotes with these features.
which one of these best defines climate
please help i will mark brainlest answer if correct asap
Answers
Answer:
Long term condition of the atmosphere
Explanation:
I think this is right.
I hope this helps! (✿◕‿◕✿)
Which statement gives an advantage of multicellular organisms?
A. Cells are able to provide support
B. They can reproduce quickly
C. Their size allows them to maintain structure easily
D. They need small amounts of energy
Answers
Answer:
A. Cells are able to provide support
In chemistry, one often uses a unit of charge known as the Faraday, F, which has the magnitude of the charge of 1 mole of electrons. How many faradays of charge does that correspond to
Answers
Answer:
1 Faraday
Explanation:
This question seeks to test proper understanding of Faraday's second law of electrolysis which states when the same quantity electricity passes through a solution of different electrolytes, the mass of the substance deposited is directly proportional to there chemical equivalence.
This law hints at the charges of ions being the chemical equivalence and also been represented as the unit for the quantity of electricity in faraday, F. Hence, a metal with the charge of 1+ can be displaced by 1F (which is equal to 96, 500 coloumbs). This charge also represents one molecule of electron, thus 1 faraday corresponds to one mole of electrons.
NOTE: It should be noted that a metal with the charge of 2+ can be displaced by 2F and so on.