Shontal compared some of the properties of a marble to a piece
of wood. She placed a marble and a piece of wood in a bucket filled
with water. Shontal observed the wood floating on top of the water,
but the marble sank to the bottom of the bucket. Which statement
best explains why the marble and piece of wood acted differently in
the water?
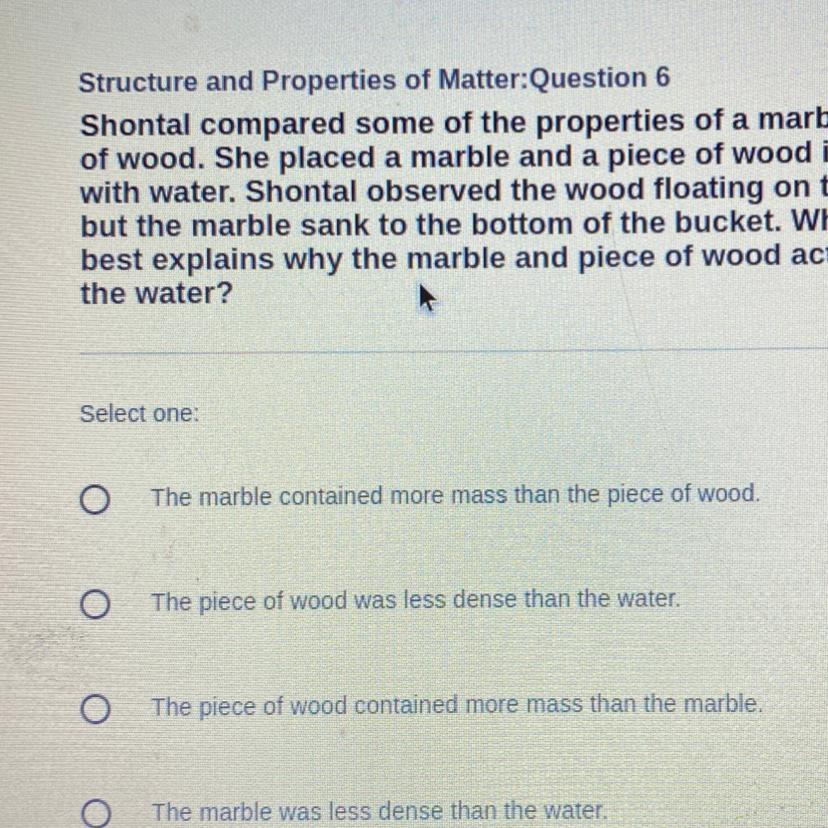
Answers
Answer:
The piece of wood was less dense than the water.
Explanation:
When an object has a lower density than water, it will float on the water.
Related Questions
If I had 50 graham cracker squares, 15 marshmallows, and 100 little
chocolate pieces, how many total s'mores could I make before I ran out of
ingredients?
Answers
What is the percent composition of each element in the compound below:
CrPO4
Answers
How are chemical reactions classified from an energy point of view?
Answers
Answer:
normanie tykohoukdh kjdj jj jjd
Explanation:
-->
Give the products of this neutralisation reaction: Sulfuric acid + magnesium hydroxide
Answers
Magnesium combined with sulfuric acid produces magnesium sulfate and hydrogen gas.
Can anyone help me with this algebraic expression
(4a+3b)² (3a-4b)²
Answers
Answer:
=144a⁴−168a³b−239a²b²+168ab³+144b⁴
Explanation:
=144a⁴−168a³b−239a²b²+168ab³+144b⁴
Answer:
[tex](4a + 3b)(4a + 3b)(3a - 4b)(3a - 4b) \\ {16a}^{2} + 12ab + {9b}^{2} + 12ab + {9a}^{2} + 12ab + 12ab + {16b}^{2} \\ {16a}^{2} + {9a}^{2} + {9b}^{2} + {16b}^{2} + 12ab + 12ab + 12ab + 12ab \\ {25a}^{2} + {25b}^{2} + 48ab[/tex]
Which of these forces does not act over a distance?
A. Friction
B. Electric
C. Magnetic
D. Gravitational
Answers
similarities of ancient and modern philosophy
Answers
Please help me with dis
Answers
Answer:
c
Explanation:
Measurements show that unknown compound X has the following composition:
element mass %
carbon 74.8%
hydrogen 25.1%
Write the empirical chemical formula of X. please help
Answers
the empirical chemical formula of X is CH4
Explanation:Step 1: Imagine you have a sample of compound weighing exactly . Multiply the mass of this sample by the mass percents to find the mass of each element in the sample
Step 2: Divide the mass of each element by the element's molar mass to find the moles of each element in the sample. Remember to round your answers to the correct number of significant digits.
Step 3: Divide the moles of each element by the the smallest number of moles of any element to find the mole ratio of elements in the sample.
Step 4: Multiply the mole ratio by the smallest whole number that changes it into a whole number ratio to find the atom ratio of elements in the sample.
Note that the result of each multiplication must equal a whole number only within measurement uncertainty.
The measurement uncertainty in this calculation comes from the measurement uncertainty of the mass percents given in the question. The mass percents each have significant digits. That means each mass percent has some measurement uncertainty in the third significant digit, and only the first two significant digits can be considered completely reliable.
In Step 1 you multiplied each mass percent by something with zero uncertainty (the exactly you assumed your sample weighed), in Step 2 you divided by a measurement with more than significant digits (the molar mass of the elements), and in Step 3 you divided by a measurement with the same number of significant digits (the least number of moles of any element in the compound). None of these steps added to the measurement uncertainty of your calculation.
Therefore, the measurement uncertainty in the final result of all your calculations is determined by the measurement uncertainty in the original mass percents and will be in the third significant digit. That means the result of each of the final multiplication steps must equal a whole number only to within the first two significant digits.
The whole numbers in the last column of the table are and .
how many molecules of NaCl are in 32.5 G
Answers
Answer :32,5 g NaCl equal 0,556 moles.
Explanation:
A gas in the form of Bubbles is released is it a chemical change or a physical change
Answers
Answer:
The formation of a gas is a clue to chemical changes. The bubbles of gas that you observed form when an antacid is dropped into water is an example of change. Another clue that a chemical change has occurred is the formation of a solid.
Explanation:
PLEASE HELP MEEE THIS IS FOR a gradE AND IF I FAIL I FaIL THIS CLASs HELP ME
Answers
Answer:
stream
Explanation:
because a stream goes into a lake
Answer:
D) Stream
Explanation:
A 2.00 g sample of ammonia (NH3) reacts with 4.00 g of oxygen (O2) according to the equation 4NH3+5O2→4NO+6H2O. How much excess reactant remains after the reaction has stopped ( knowing that NH3 is the excess reactant )?
Answers
Answer:
0.017 moles of ammonia remains after the reaction is stopped.
Explanation:
The reaction is:
4NH₃ + 5O₂ → 4NO + 6H₂O
The first step is to convert the mass to moles, of each reactant:
2 g . 1mol/ 17g = 0.117 moles of NH₃
4 g . 1mol /32g = 0.125 moles of O₂
Ammonia, states the question, is the excess reactant so we can confirm it,
5 moles of oxygen need 4 moles of ammonia to react (by stoichiometry)
Then, 0.125 moles of oxygen may react to (0.125 . 4) / 5 = 0.1 moles
As we have 0.117 moles of ammonia and we need 0.1 moles.
(0.117 - 0.1) = 0.017 moles remains after the reaction is completed.
If we convert the moles to mass we have:
0.017 mol . 17 g /1mol = 0.289 g
An atom of an element always contains
Answers
Answer:
An atom of an element always contain three fundamental particles called electrons(e-) , neutron ( n zero) and protons ( p+)
An atom of an element always contains a proton, electron and neutron.
What is an atom ?Every atom is made up of a nucleus and one or more electrons that are attached to the nucleus. One or more protons and a number of neutrons make up the nucleus. Only the most common type of hydrogen is neutron-free. Every solid, liquid, gas, and plasma is made up of atoms that are either neutral or ionized.
But when it comes to the word atom, we must go back to 400 B.C. Greece. And Democritus, a brilliant philosopher, proposed the Greek word atomos, which means uncuttable.
Every atom is made up of a nucleus and one or more electrons that are attached to the nucleus. One or more protons and a number of neutrons make up the nucleus.
Thus, An atom of an element always contains a proton, electron and neutron.
To learn more about an atom, follow the link;
https://brainly.com/question/29695801
#SPJ2
How many moles of carbon dioxide are produced when 5.12 moles of glucose undergoes a combustion reaction?
Answers
Answer: 30.72 moles of [tex]CO_2[/tex] will be produced from 5.12 moles of glucose.
Explanation:
The balanced chemical equation for combustion of glucose is:
[tex]C_6H_{12}O_6+6O_2\rightarrow 6CO_2+6H_2O[/tex]
According to stoichiometry :
1 moles of glucose produce = 6 moles of [tex]CO_2[/tex]
Thus 5.12 moles of glucose produce =[tex]\frac{6}{1}\times 5.12=30.72moles[/tex] of [tex]CO_2[/tex]
Thus 30.72 moles of [tex]CO_2[/tex] will be produced from 5.12 moles of glucose.
please help! Will give Brainliest!
Answers
Answer: the answer is c
Explanation:
also, is that canvas? sajdasds
Think of the of the body that we need in order to do things like walk, run etc. What systems are these? How do they work together so you can do these activities?
Answers
Answer:
I dont get this
Explanation:
What are the charges on ions of Group 1A, Group 3A (aluminum), and Group 5A?
Answers
Answer:
Group 1A: 1+
Group 3A: 3+
Group 5A: 3+ or 5+
Explanation:
The chemical equation, Cr + Fe(NO3)2 → Fe + Cr(NO3)3, is an example of which type of reaction?
Answers
Answer:
Redox type
Explanation:
The reaction is:
2Cr + 3Fe(NO₃)₂ → 2Fe + 2Cr(NO₃)₃
2 moles of chromium can react to 3 moles of iron (II) nitrate in order to produce 2 moles of iron and 2 moles of chromium nitrate.
If we see oxidation state, we see that chromium changes from 0 to +3
Iron changed the oxidation state from +2 to 0
Remember that elements at ground state has 0, as oxidation state.
Iron is being reduced while chromium is oxidized. Then, the half reactions are:
Fe²⁺ + 2e⁻ ⇄ Fe (Reduction)
Cr ⇄ Cr³⁺ + 3e⁻ (Oxidation)
When an element is being reduced, while another is being oxidized, we are in prescence of a redox reaction.
If combining cation A with a +3 charge and anion X with a -3 charge, how would we write the compound?
O AX
O A3X3
O 3AX
Answers
Answer: it’s not A3X3
Explanation:
1. AX
For binary ionic compounds (ionic compounds that contain only two types of elements), the compounds are named by writing the name of the cation first followed by the name of the anion.If a cation A carries +3 charge and an anion X carries -3 charge, the compound so formed is AX. Since the charges gets cancelled out.For example: consider aluminum phosphate, aluminum carries +3 charge and phosphate carries -3 charge, the molecular formula for the compound will be AlPO₄.Learn more:
brainly.com/question/11164440
HELP PLEASE
What does the 2 mean in the formula of calcium nitrate? What does the 3
mean?
Answers
Answer:
Calcium nitrate is an inorganic compound with the chemical formula Ca(NO3)2. ... It is a nitrate salt of Calcium which contains calcium and nitrogen and oxygen. Calcium nitrate is a white or whitish-grey coloured granular solid which absorbs moisture from the air and is usually found as a tetrahydrate compound Ca(NO3)2.
Explanation:
1. If you have 5g of pennies how many dozen pennies do you have?
Answers
Answer:
15.69 dozen
Explanation:
Mass of penny = 5 g
Dozens of penny =..?
Next, we shall convert 5 g to gross. This can be obtained as follow:
3824 g = 1000 gross
Therefore,
5 g = 5 g × 1000 gross / 3824 g
5 g = 1.3075 gross
Thus, 5 g is equivalent to 1.3075 gross.
Finally, we convert 1.3075 gross to dozen. This can be obtained as follow:
1 gross = 12 dozen
Therefore,
1.3075 gross = 1.3075 gross × 12 dozen / 1 gross
1.3075 gross = 15.69 dozen
Thus, 5 g of penny is equivalent to 15.69 dozen
What are the reactants in the following equation; NaOH + HCI --> NaCl + H2O
a)NaOH and HCI
b)HCI and H20
c) NaOH and NaCl
d)HCI and water
Answers
Which date has the longest daylight and the shortest darkness
Answers
The Winter Solstice.
A balloon occupies 175 cm at 25.0 C. What volume will it occupy at 42.5 °C?
Answers
175 cm³ instead of 175 cm.
Answer:
297.5 cm³
Explanation:
From the question;
Initial volume; V1 = 175 cm³
Initial temperature; T1 = 25°C
Final temperature; T2 = 42.5°C
From Charles law we can find the volume V2 from the equation;
V1/T1 = V2/T2
Making V2 the subject gives;
V2 = V1 × T2/T1
V2 = (175 × 42.5)/25
V2 = 297.5 cm³
what is the meaning of flourish?
Answers
*For Example* :
"wild plants flourish on the banks of the lake"
Can you bond 2 silicon atoms (Si) together to form a complete (real) molecule? Explain your reasoning
Answers
Answer: no
Explanation: because silicon cannot share over 3 bonds
Rubidium (Rb) has two isotopes. Using the data in the table below,
calculate the relative atomic mass of Rb. Give your answer to 3 significant
figures.
Answers
Answer:
85.6
Explanation:
(72 x 85) + (28 x 87)
-----------------------------
100
= 85.56
= 85.6 (3 s.f.)
the indicated gene codes for a protein made up of the amino acid
Answers
Answer:
N/A
Explanation:
Is this a true or false answer or.....?
Name the following cycloalkane:
CH2CH2CH2CH3
A. butylcyclohexane
B. butylcyclopentane
C. propylcyclohexane
Answers
Answer:
The answer for this question is A. butylcyclohexane
Explanation:
Choose letter A
You welcome have a nice day.
Name of the given compound is butyl cyclohexane.
What are cyclo alkanes?Cyclo alkanes are those organic compounds in which alkanes are present in the cyclic form.
In the nomenclature of cyclo alkane, name of the substitute is put before the name of the main ring. In the given structure number of carbons in ring is 6 so that given cycloalkane is cyclohexane and butyl group is attached as substitute. So name of the given compound is butyl cyclohexane.
Butyl cyclopentane is wrong because 6 carbons are present in ring.Propylcyclohexane is also wrong because 4 carbons are attached in the ring.Hence name of the compound is butyl cyclohexane.
To know more about cycloalkanes, visit the below link:
https://brainly.com/question/25058711
#SPJ2